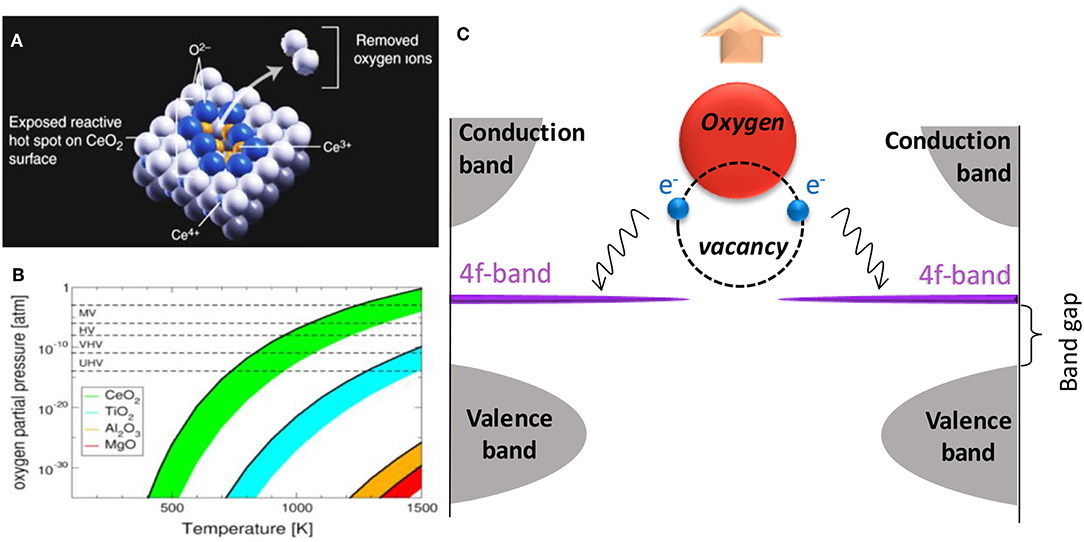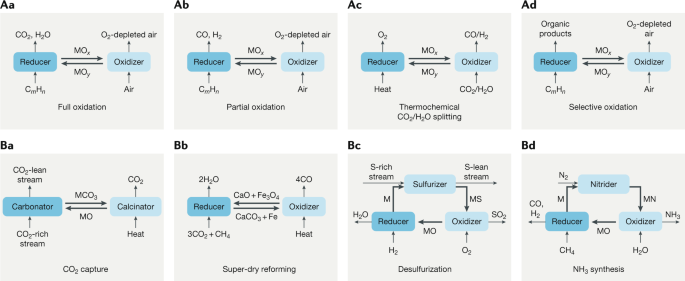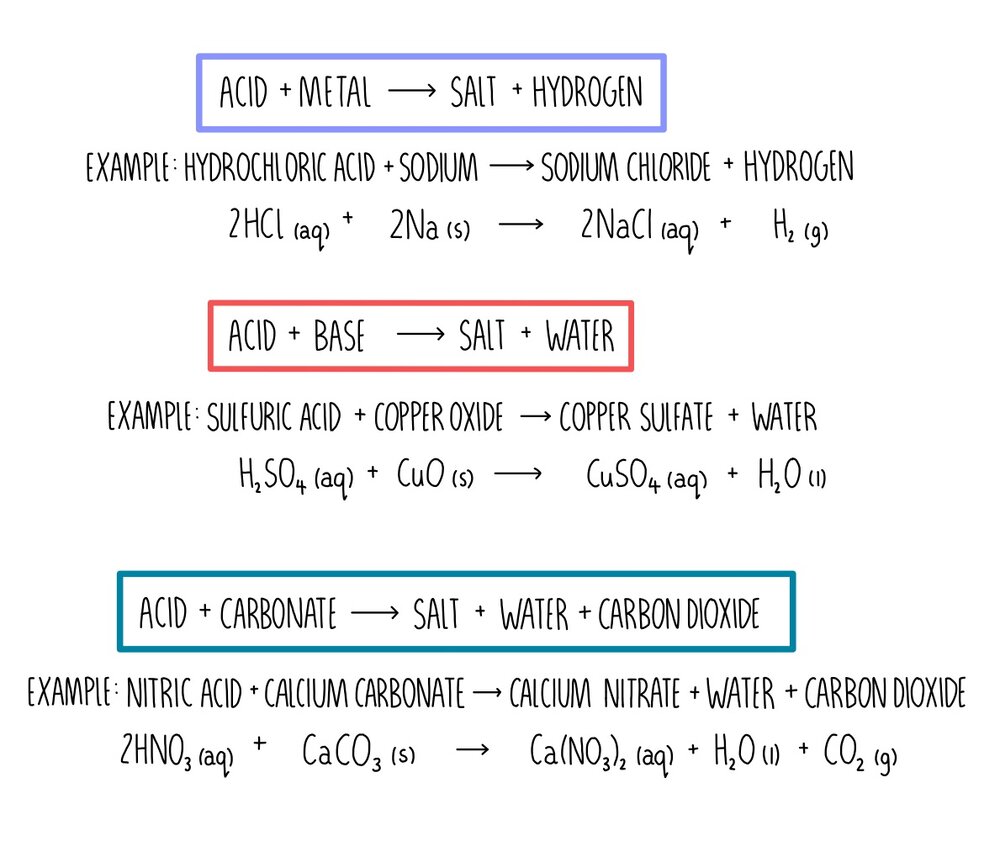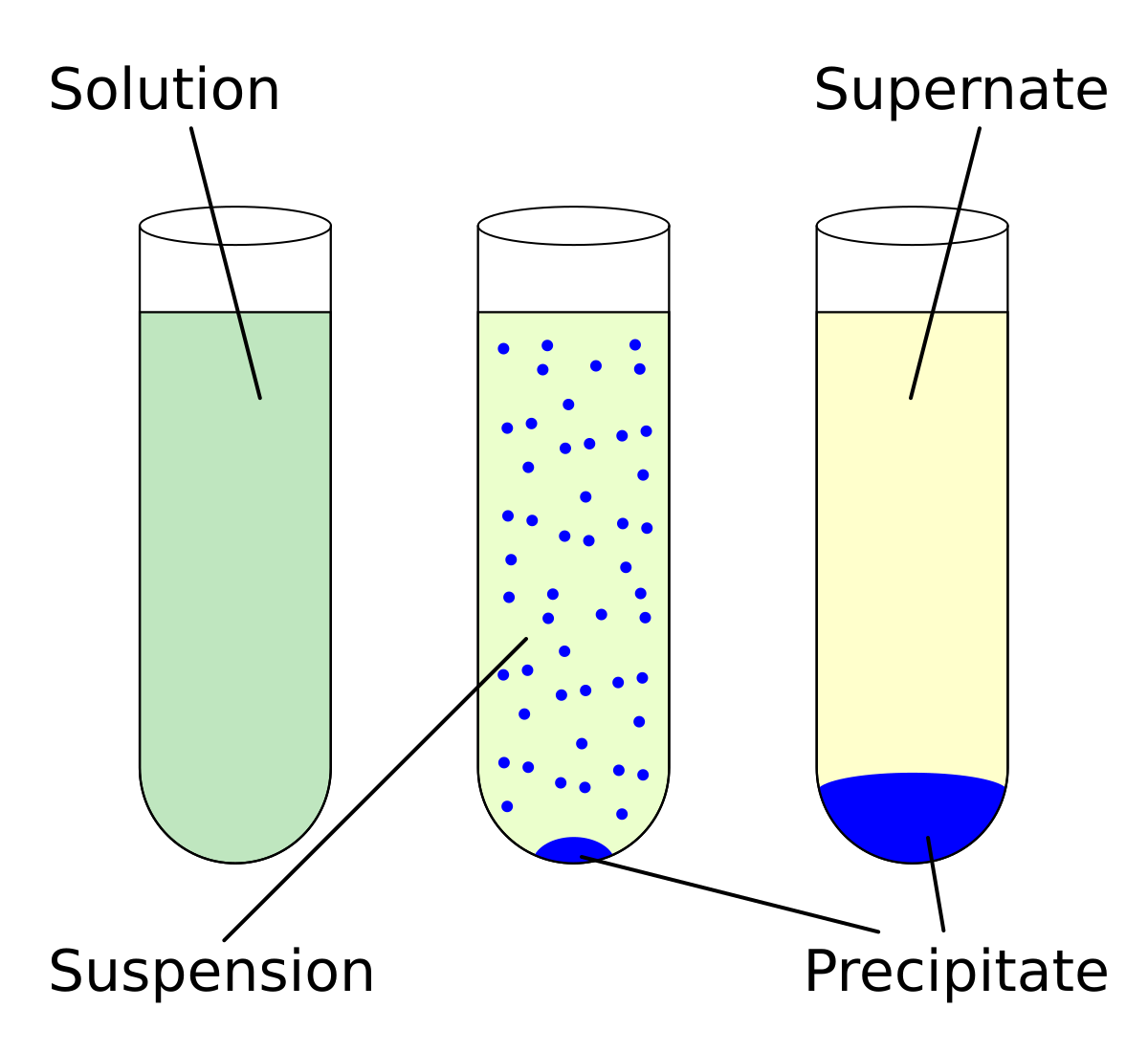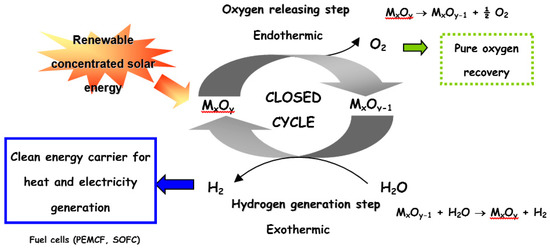
ChemEngineering | Free Full-Text | Metal Oxides Applied to Thermochemical Water-Splitting for Hydrogen Production Using Concentrated Solar Energy
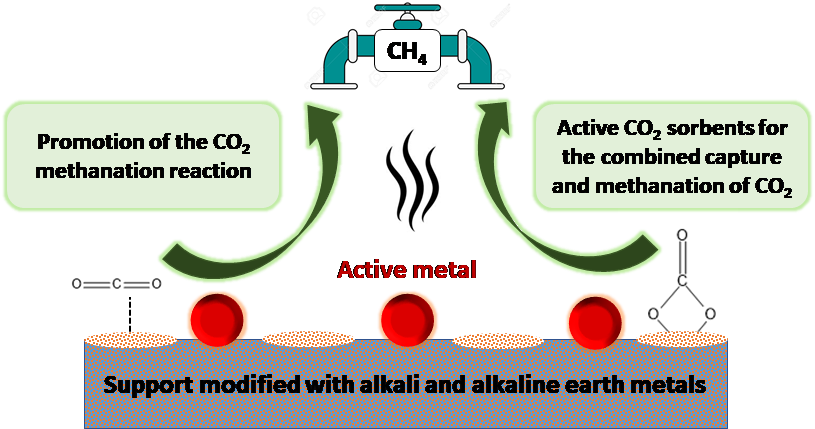
Catalysts | Free Full-Text | The Role of Alkali and Alkaline Earth Metals in the CO2 Methanation Reaction and the Combined Capture and Methanation of CO2

When alkali metals are heated in excess of air. What is the nature of oxides formed? - CBSE Class 11 Chemistry - Learn CBSE Forum

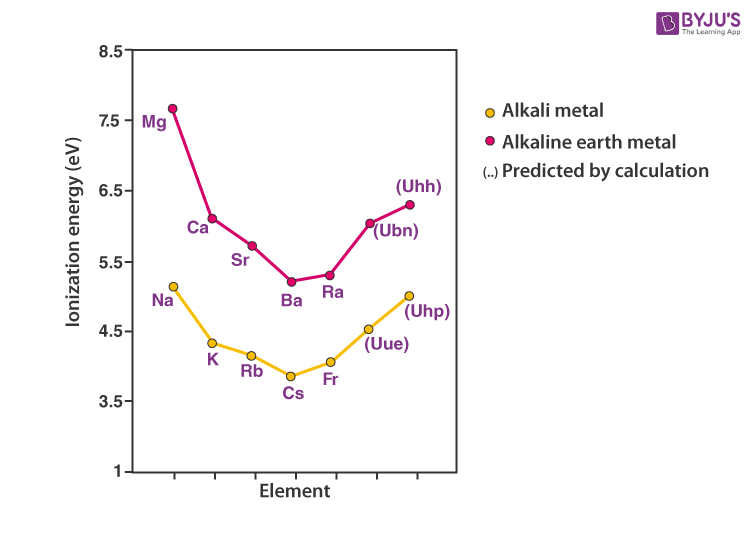
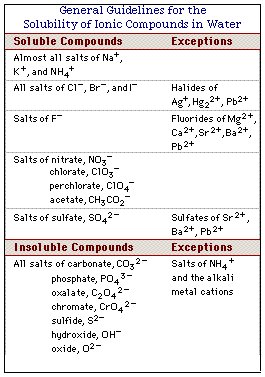
Compare the alkali metals and alkaline earth metals with respect to (i) ionisation enthalpy (ii) basicity of oxides and (iii) solubility of hydroxides.

%20with%20water.jpg?ezimgfmt=rs:363x250/rscb1/ngcb1/notWebP)