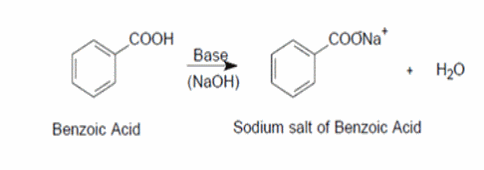
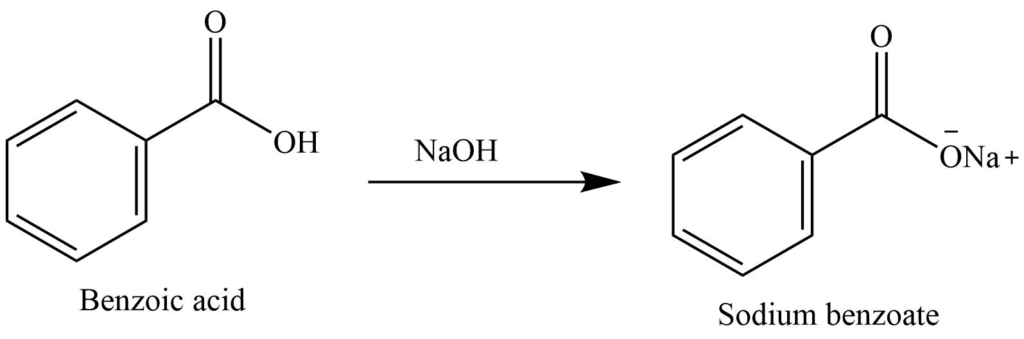
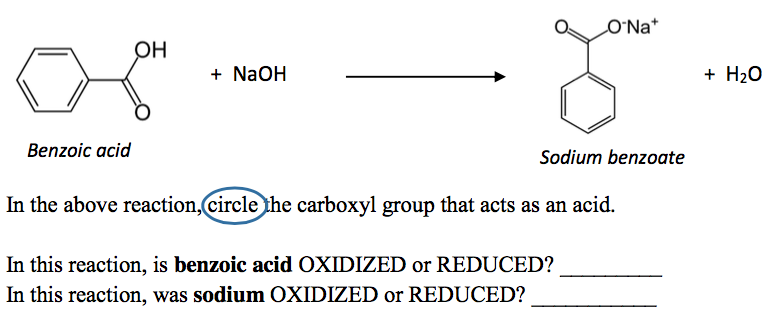1. Draw a balanced chemical equation for the reaction that would occur between benzoic acid and aqueous sodium hydroxide. 2. Draw a balanced chemical equation for the reaction that would occur between

Draw the products of benzoic acid reacting with sodium hydroxide. Draw the products of the pyridine reacting with hydrochloric acid. Use the "+/-" button to add the charge (and H atom).
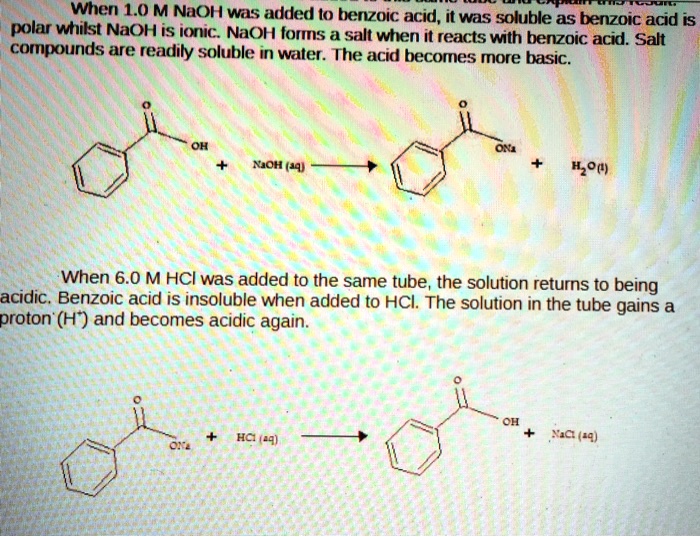
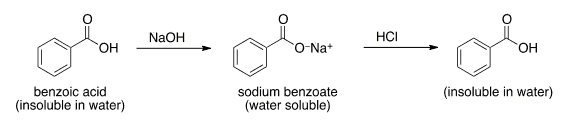
SOLVED: When 1.0 M NaOH was added (0 benzoic acid, it was soluble as benzoic acid is polar wtitst NaOH is ioric; NaOH forms salt when i reacts with berizoic aczo Sad

Experiment 6 Extraction - Separation of Benzoic Acid and Phenanthrene - Experiment 6: Extraction - - Studocu

Titration curves of benzoic acid for different analyte concentrations... | Download Scientific Diagram

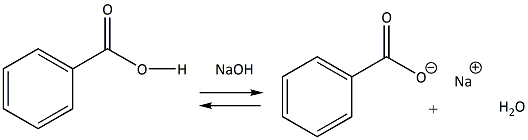
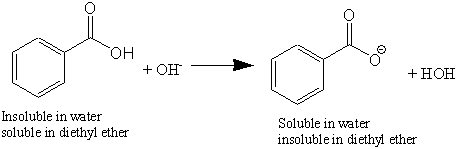
Write a balanced equation for the reaction of benzoic acid with hydroxide ion. Why is it necessary to extract the ether layer with sodium hydroxide? | Homework.Study.com
✓ Solved: Explain the results for the tube in which 1.0 M NaOH was added to benzoic acid. Write an equation...
![When a solution of benzoic acid was titrated with NaOH the pH of the solution when half the acid neutralized was 4.3 . Dissociation constant of the acid is [Use log 2 = 0.3] . When a solution of benzoic acid was titrated with NaOH the pH of the solution when half the acid neutralized was 4.3 . Dissociation constant of the acid is [Use log 2 = 0.3] .](https://haygot.s3.amazonaws.com/questions/1835171_829774_ans_40c76abc129644cfaff5cbb818338569.png)
When a solution of benzoic acid was titrated with NaOH the pH of the solution when half the acid neutralized was 4.3 . Dissociation constant of the acid is [Use log 2 = 0.3] .

Write a chemical equation that explains the observations on the addition of HCL to the mixture of benzamide and NaOH subsequent to heating. | Homework.Study.com

Explain the results for the tube in which 1.0 m naoh was added to benzoic acid. write an equation for this, - Brainly.com
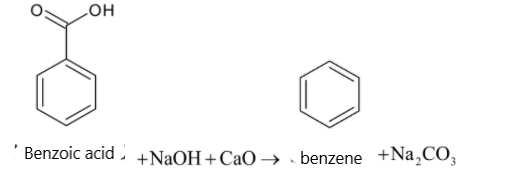
Heating of carboxylic acid with soda lime results in:A. dehydrationB. dehydrogenationC. decarboxylationD. addition of ${{{O}}_2}$

Crystals | Free Full-Text | Co-Crystallization Kinetics of 2:1 Benzoic Acid–Sodium Benzoate Co-Crystal: The Effect of Templating Molecules in a Solution