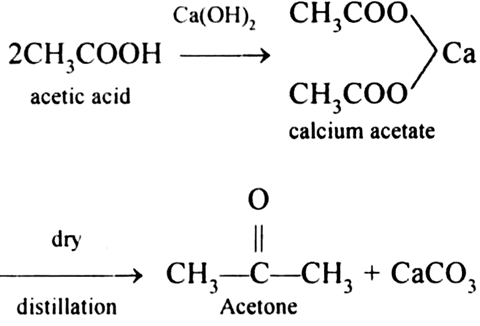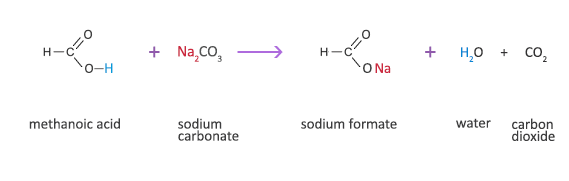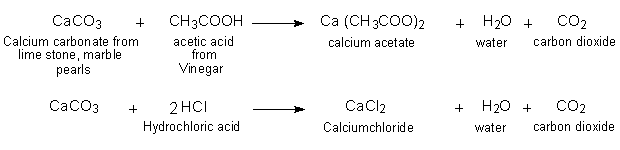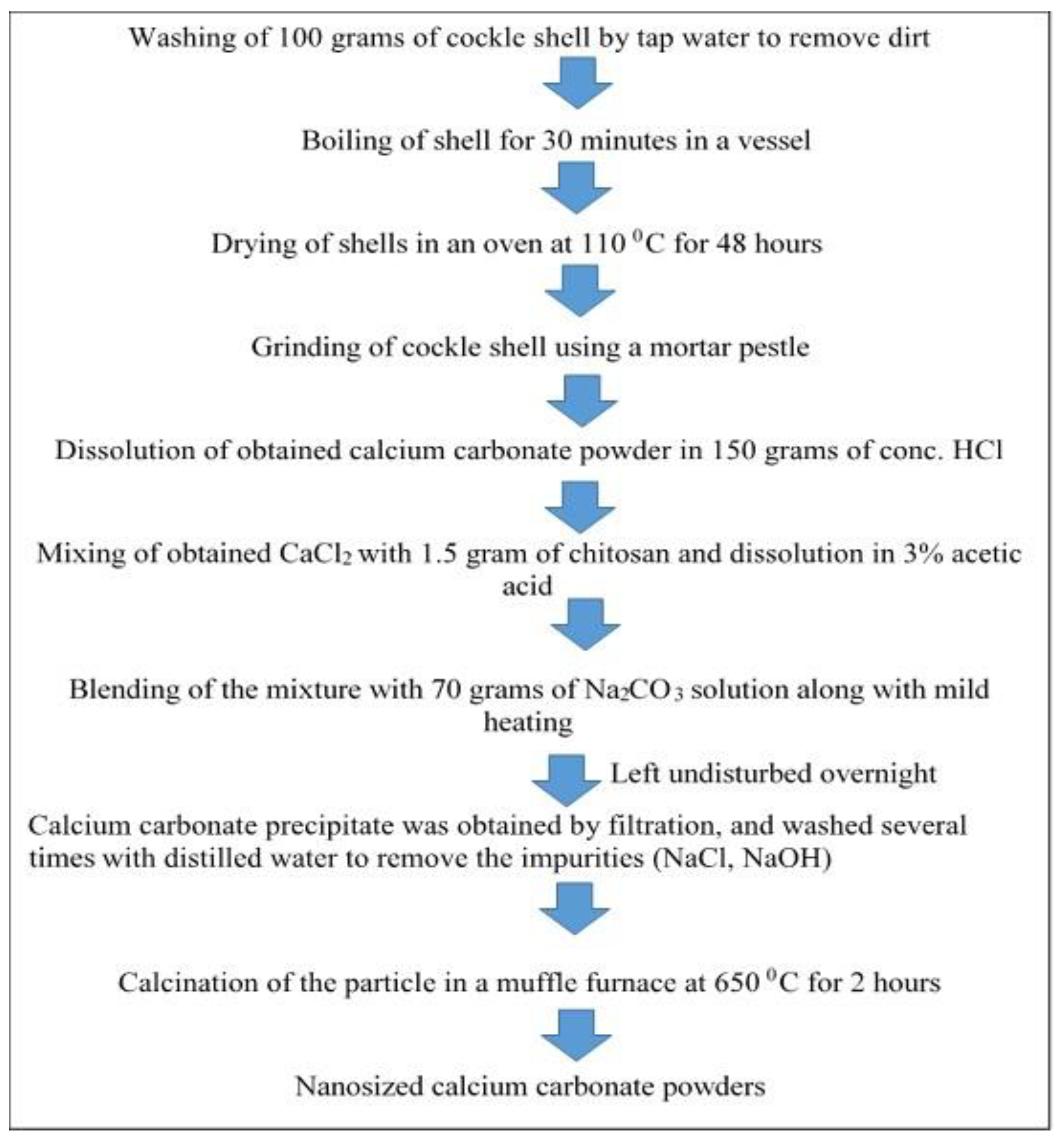
Applied Sciences | Free Full-Text | The Processing of Calcium Rich Agricultural and Industrial Waste for Recovery of Calcium Carbonate and Calcium Oxide and Their Application for Environmental Cleanup: A Review

SEM images of the synthesized calcium carbonate by the extraction of... | Download Scientific Diagram
![PDF] Dissolution of steelmaking slags in acetic acid for precipitated calcium carbonate production | Semantic Scholar PDF] Dissolution of steelmaking slags in acetic acid for precipitated calcium carbonate production | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2d4d58751a65ed0fe044f73117db0550773b0abf/4-Figure1-1.png)
PDF] Dissolution of steelmaking slags in acetic acid for precipitated calcium carbonate production | Semantic Scholar
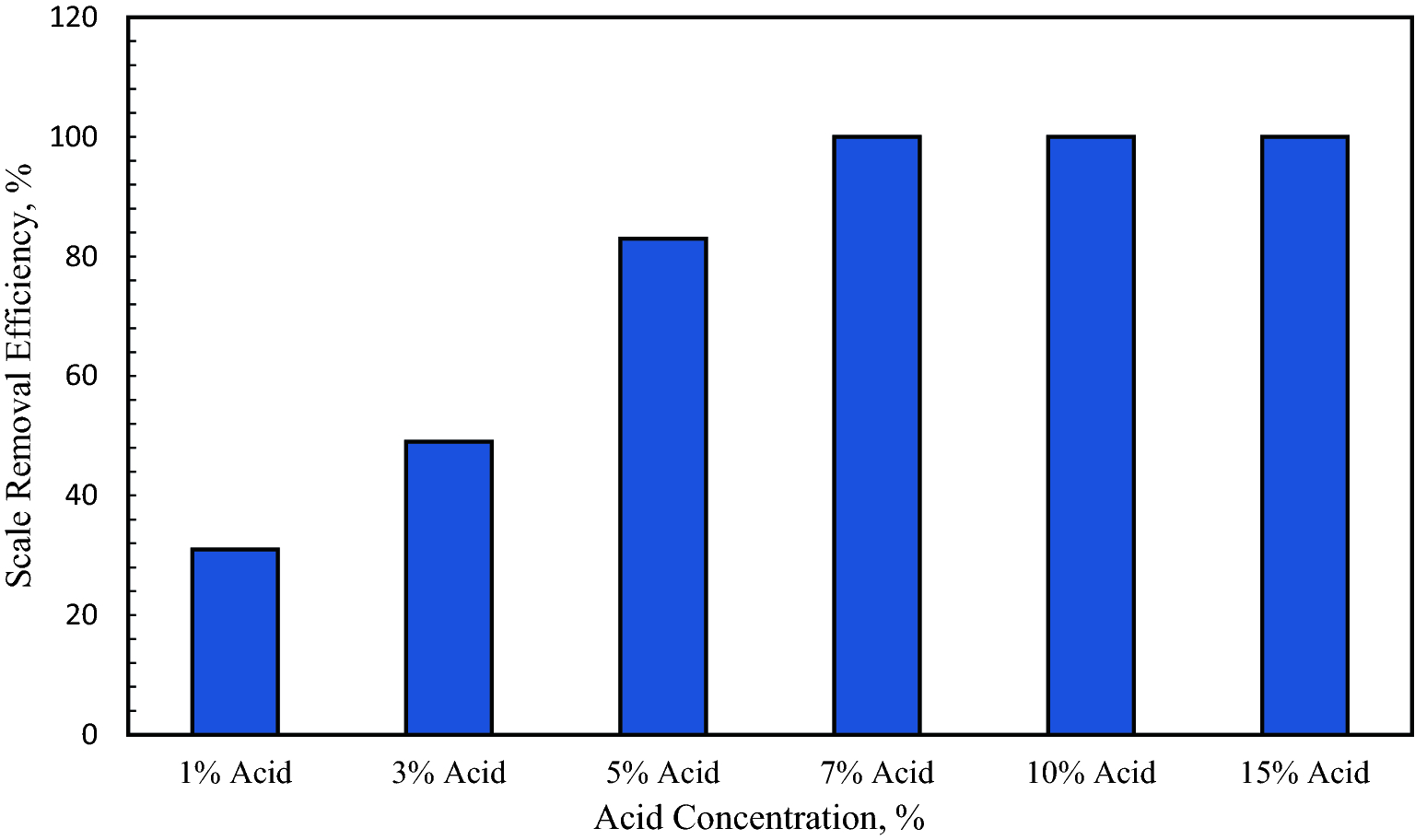
Single step calcium sulfate scale removal at high temperature using tetrapotassium ethylenediaminetetraacetate with potassium carbonate | Scientific Reports
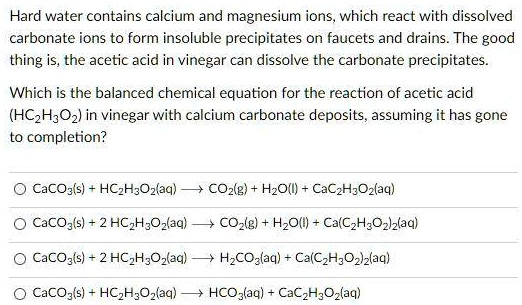
SOLVED: Hard water contains calcium and magnesium ions, which react with dissolved carbonate ions to form insoluble precipitates on faucets and drains. The good thing is, the acetic acid in vinegar can
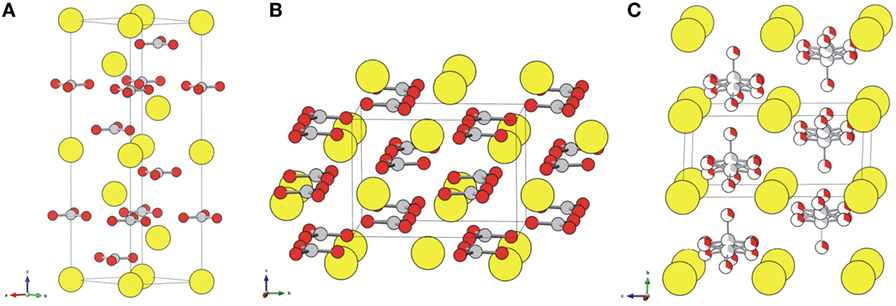
Frontiers | Calcium Carbonate Precipitation for CO2 Storage and Utilization: A Review of the Carbonate Crystallization and Polymorphism

SOLVED: Pearls are built from calcite and aragonite, essentially two forms of calcium carbonate CaCO3. They disintegrate in vinegar, which contains acetic acid. Formulate the net ionic equation for this process. [note: