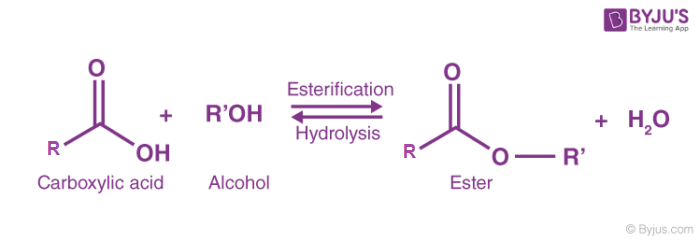
How can you convert a carboxylic acid into an ester? a. Heat with an alcohol and catalytic acid. b. Deprotonate with a base and react with an alcohol. c. Deprotonate with a

Oxidation of alcohols to carboxylic acids with polymer-supported TEMPO. | Download Scientific Diagram
![write structures for the following carboxylic acids and alcohols that will be used in this laboratory. [{Image src='lab3603143357806879253.jpg' alt='lab' caption=''}] | Homework.Study.com write structures for the following carboxylic acids and alcohols that will be used in this laboratory. [{Image src='lab3603143357806879253.jpg' alt='lab' caption=''}] | Homework.Study.com](https://homework.study.com/cimages/multimages/16/structures_of_molecules_acids_and_alcohols1372292982843607771.png)
write structures for the following carboxylic acids and alcohols that will be used in this laboratory. [{Image src='lab3603143357806879253.jpg' alt='lab' caption=''}] | Homework.Study.com
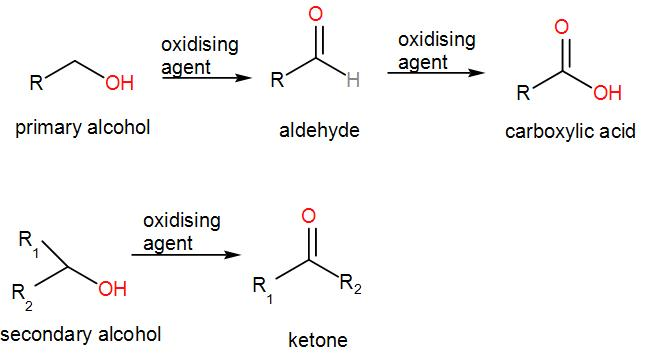
Carboxylic acids can be prepared from alcohols, nitriles or esters. Which statements are correct?1. Both primary and secondary alcohols can be oxidised to carboxylic acids.2. Carboxylic acids can be made from nitriles

Question Video: Determining the Name of the Ester Produced from the Esterification of Propanoic Acid with Ethanol | Nagwa

Hydroxymethylaniline Photocages for Carboxylic Acids and Alcohols | The Journal of Organic Chemistry


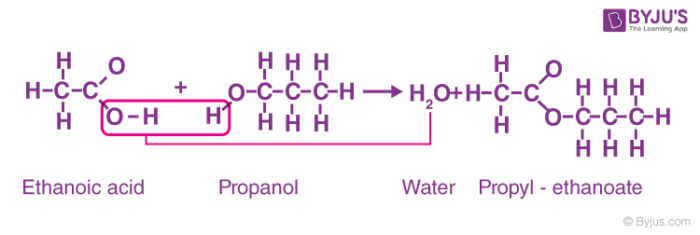
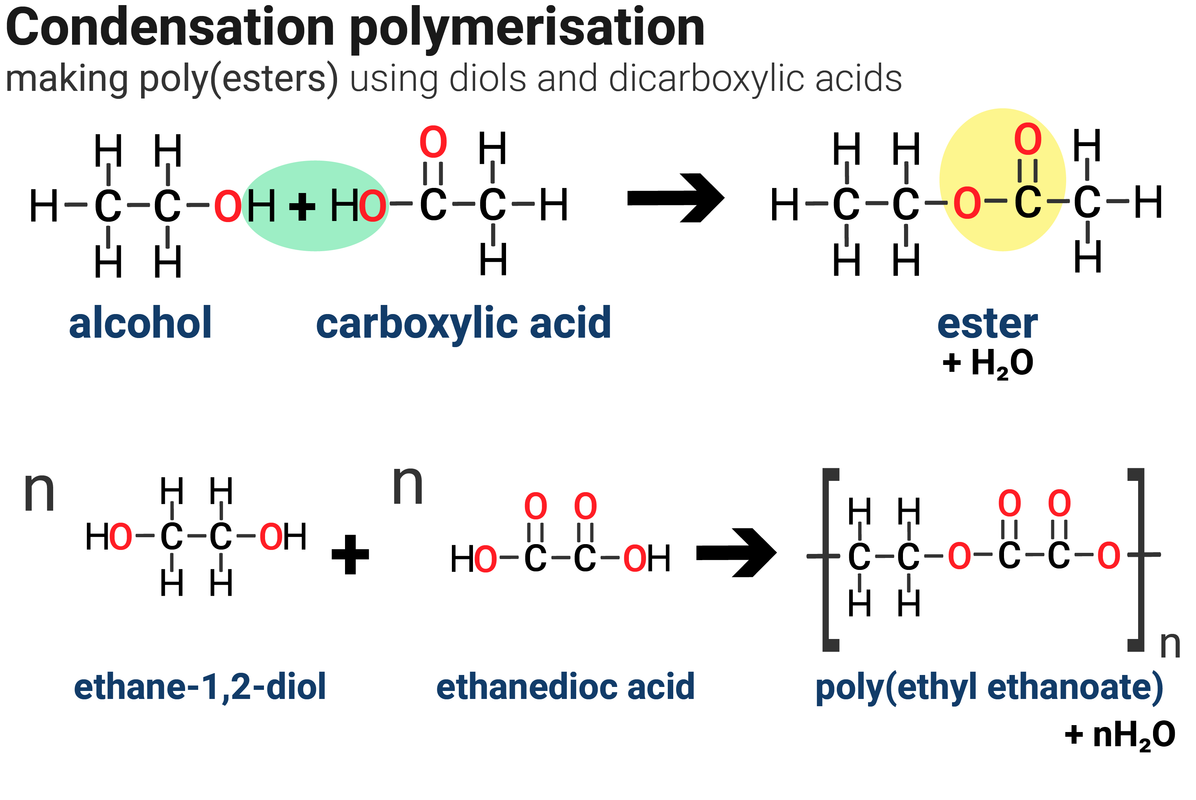
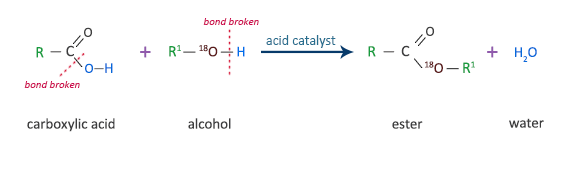


![Carboxylic acids [O] [O] 1o alcohol aldehyde carboxylic acid methanol - ppt download Carboxylic acids [O] [O] 1o alcohol aldehyde carboxylic acid methanol - ppt download](https://slideplayer.com/slide/14476413/90/images/6/Reactions+nucleophilic+substitution+condensation+reaction+reverse+%3D.jpg)




![Oxidation to carboxylic acids [H2CrO4 + others] - ChemistryScore Oxidation to carboxylic acids [H2CrO4 + others] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Oxidation-to-carboxylic-acids-H2CrO41-768x327.png)