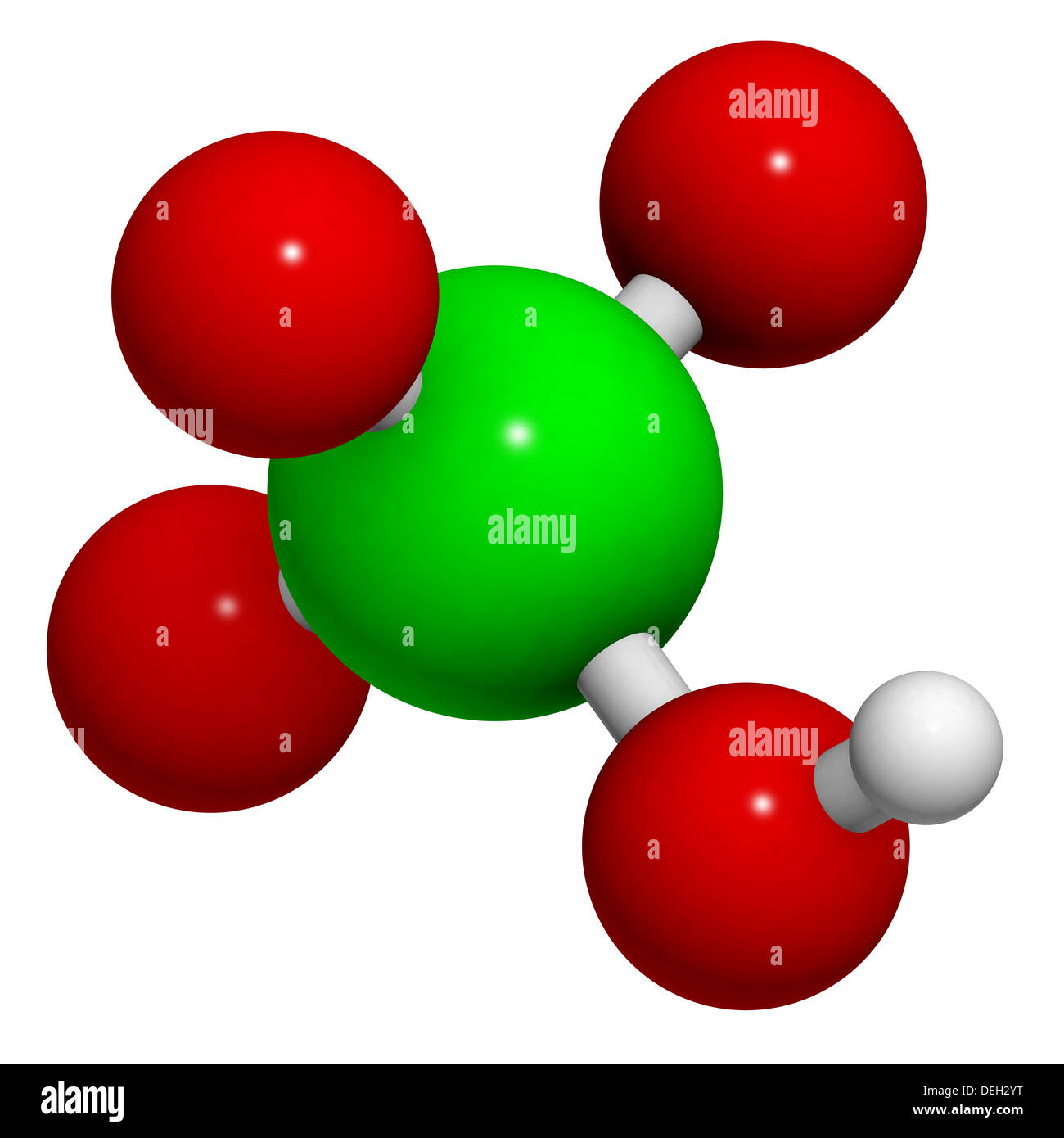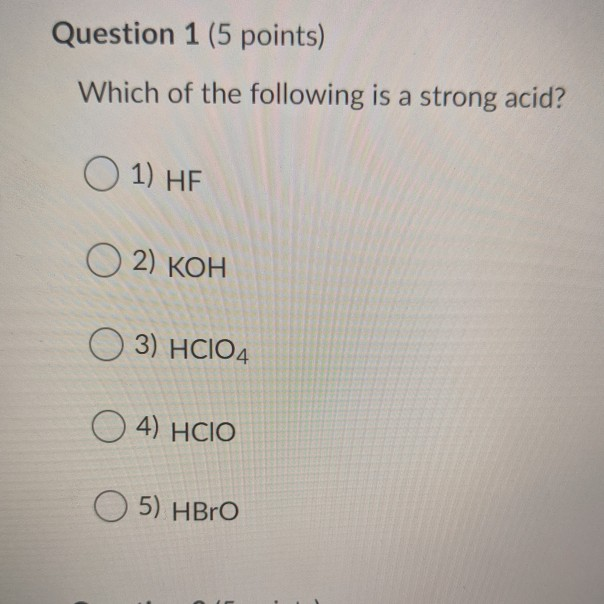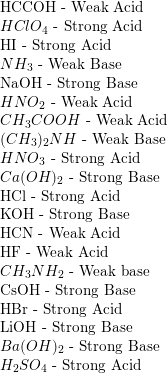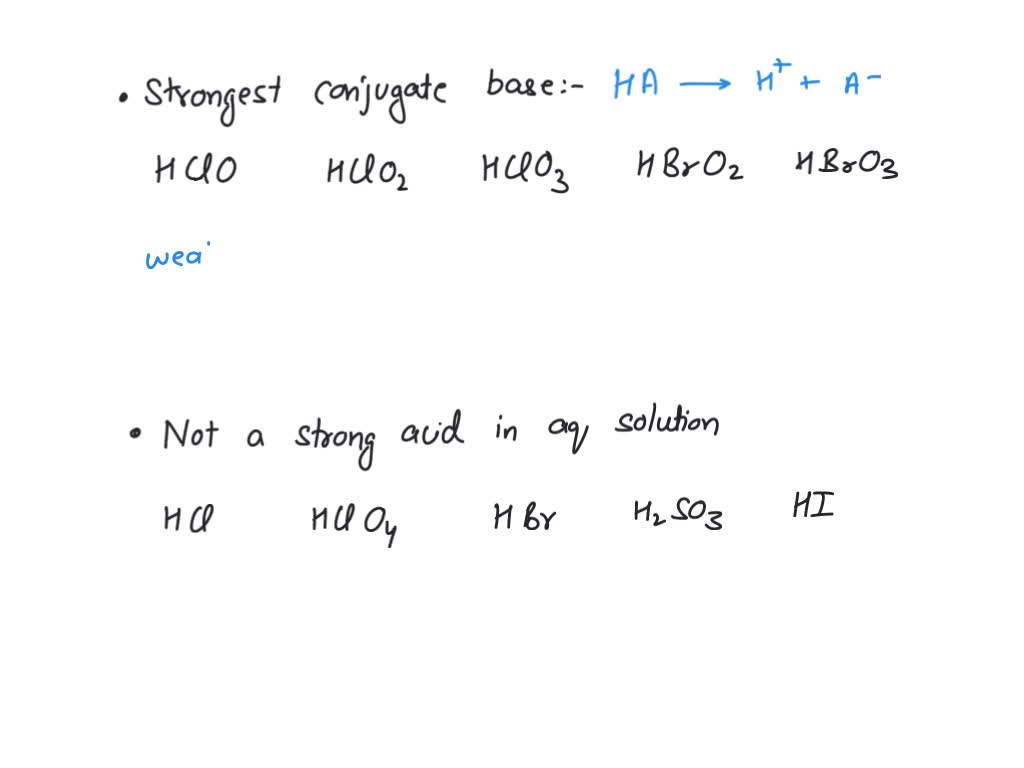
SOLVED: in the following list, which one is considered a conjugate base of a weak acid Select one: a. HClO4 b. HCO2H c. CH3NH2 d. Ba(OH)2 e. CH3CO2- 2) in the following
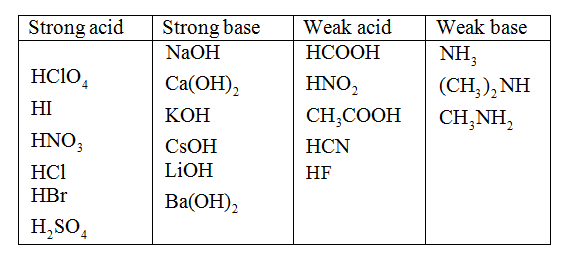
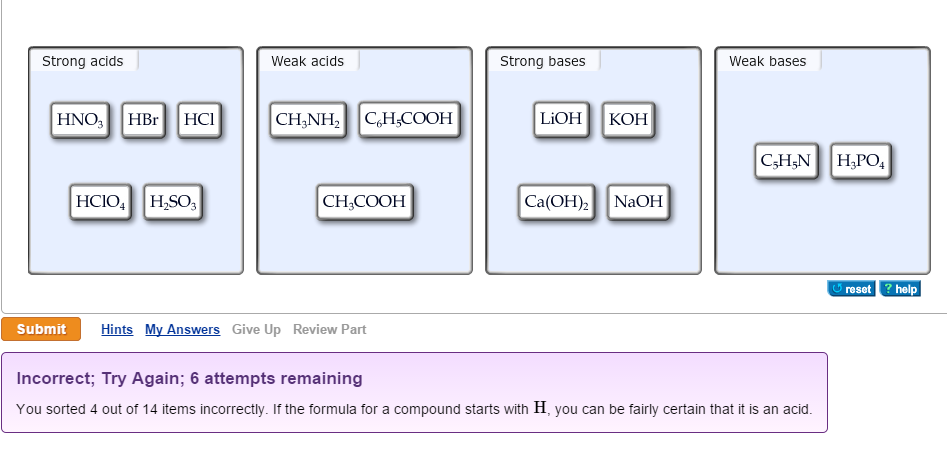
Classify each substance as a strong acid, strong base, weak acid, or weak base - Home Work Help - Learn CBSE Forum

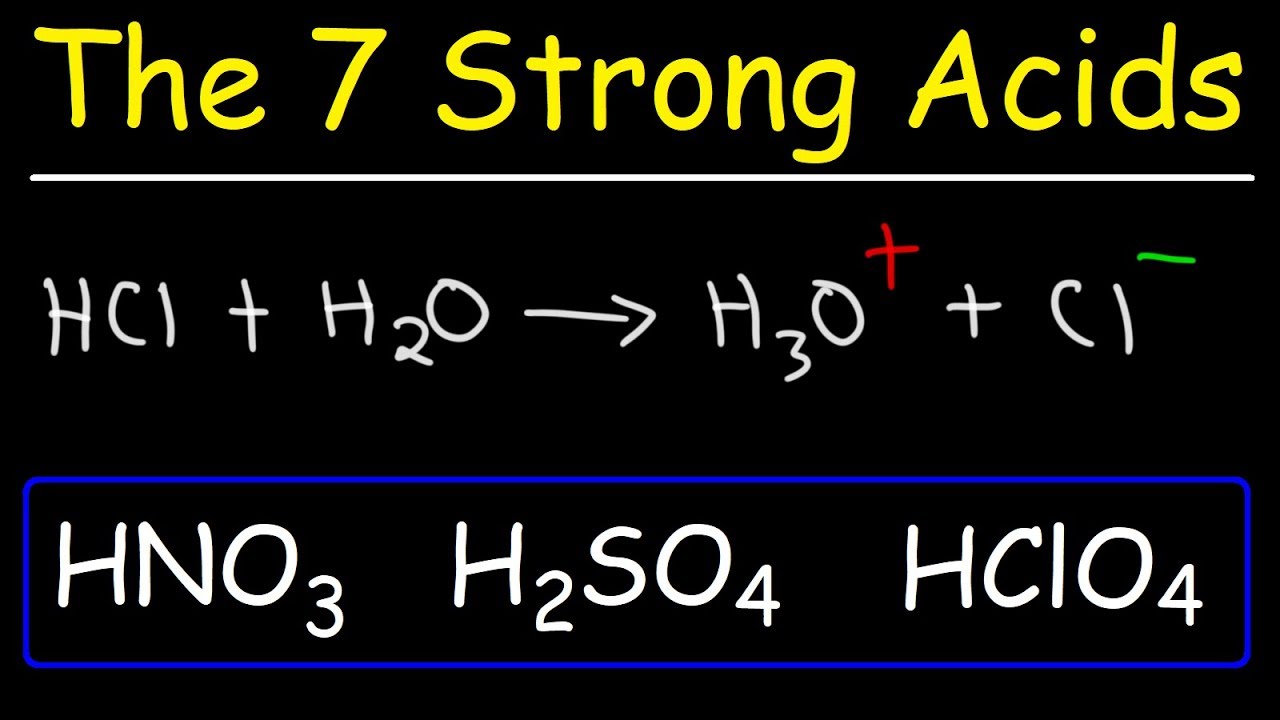
HClO_(4),HNO_(3)` and `HCl` are all strong acids in aqueous solution.In glacial acetic acid medium, - YouTube