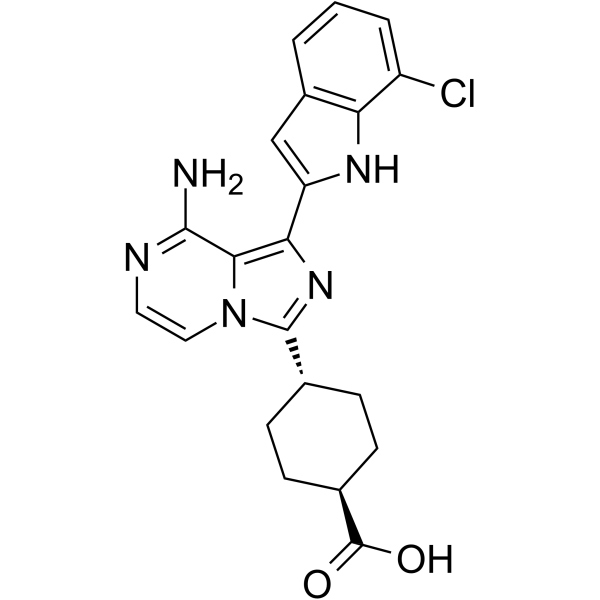
Stereoselective Syntheses of 4-Oxa Diaminopimelic Acid and Its Protected Derivatives via Aziridine Ring Opening | Organic Letters
![1-[4-(benzyloxy)-1-oxa-9-azaspiro[5.5]undec-9-yl]-3-methyl-1-butanone SDF/Mol File - C21H31NO3 - Over 100 million chemical compounds | Mol-Instincts 1-[4-(benzyloxy)-1-oxa-9-azaspiro[5.5]undec-9-yl]-3-methyl-1-butanone SDF/Mol File - C21H31NO3 - Over 100 million chemical compounds | Mol-Instincts](https://static.molinstincts.com/compound_common/1-4-benzyloxy-1-oxa-9-azaspiro-5-5-undec-9-yl-3-methyl-1-butanone-2D-structure-CT1101592826.png)
1-[4-(benzyloxy)-1-oxa-9-azaspiro[5.5]undec-9-yl]-3-methyl-1-butanone SDF/Mol File - C21H31NO3 - Over 100 million chemical compounds | Mol-Instincts
![3R)-N-[(2R)-butan-2-yl]-8-methyl-4-propanoyl-1-oxa-4-azaspiro[4.5]decane-3-carboxamide SDF/Mol File - C17H30N2O3 - Over 100 million chemical compounds | Mol-Instincts 3R)-N-[(2R)-butan-2-yl]-8-methyl-4-propanoyl-1-oxa-4-azaspiro[4.5]decane-3-carboxamide SDF/Mol File - C17H30N2O3 - Over 100 million chemical compounds | Mol-Instincts](https://static.molinstincts.com/compound_common/3R-N-2R-butan-2-yl-8-methyl-4-propanoyl-1-oxa-4-azaspiro-4-5-decane-3-2D-structure-CT1069058976.png)
3R)-N-[(2R)-butan-2-yl]-8-methyl-4-propanoyl-1-oxa-4-azaspiro[4.5]decane-3-carboxamide SDF/Mol File - C17H30N2O3 - Over 100 million chemical compounds | Mol-Instincts
![8-(propan-2-yl)-1-oxa-2-azaspiro[4.5]dec-2-ene SDF/Mol File - C11H19NO - Over 100 million chemical compounds | Mol-Instincts 8-(propan-2-yl)-1-oxa-2-azaspiro[4.5]dec-2-ene SDF/Mol File - C11H19NO - Over 100 million chemical compounds | Mol-Instincts](https://static.molinstincts.com/compound_common/8-propan-2-yl-1-oxa-2-azaspiro-4-5-dec-2-ene-2D-structure-CT1098932079.png)
8-(propan-2-yl)-1-oxa-2-azaspiro[4.5]dec-2-ene SDF/Mol File - C11H19NO - Over 100 million chemical compounds | Mol-Instincts
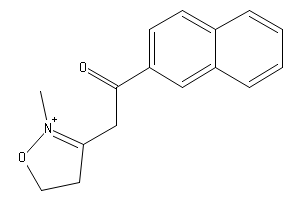
2-(2-Methyl-1-oxa-2-azonia-2-cyclopentene-3-ylacetyl)naphthalene | Chemical Substance Information | J-GLOBAL
![Highly efficient construction of an oxa-[3.2.1]octane-embedded 5–7–6 tricyclic carbon skeleton and ring-opening of the bridged ring via C–O bond cleav ... - RSC Advances (RSC Publishing) DOI:10.1039/D2RA01315K Highly efficient construction of an oxa-[3.2.1]octane-embedded 5–7–6 tricyclic carbon skeleton and ring-opening of the bridged ring via C–O bond cleav ... - RSC Advances (RSC Publishing) DOI:10.1039/D2RA01315K](https://pubs.rsc.org/image/article/2022/RA/d2ra01315k/d2ra01315k-s5_hi-res.gif)
Highly efficient construction of an oxa-[3.2.1]octane-embedded 5–7–6 tricyclic carbon skeleton and ring-opening of the bridged ring via C–O bond cleav ... - RSC Advances (RSC Publishing) DOI:10.1039/D2RA01315K
Whole genome sequencing characterization of Slovenian carbapenem-resistant Klebsiella pneumoniae, including OXA-48 and NDM-1 producing outbreak isolates | PLOS ONE

Direct Detection of OXA-48 Carbapenemase Gene in Lysate Samples through Changes in Mechanical Properties of DNA Monolayers upon Hybridization | Analytical Chemistry
Crystal Structure of OXA-58 with the Substrate-Binding Cleft in a Closed State: Insights into the Mobility and Stability of the OXA-58 Structure | PLOS ONE

OXA Flat accounting logo design on white background. OXA creative initials Growth graph letter logo concept. OXA business finance logo design. Stock Vector | Adobe Stock
![Highly efficient construction of an oxa-[3.2.1]octane-embedded 5–7–6 tricyclic carbon skeleton and ring-opening of the bridged ring via C–O bond cleav ... - RSC Advances (RSC Publishing) DOI:10.1039/D2RA01315K Highly efficient construction of an oxa-[3.2.1]octane-embedded 5–7–6 tricyclic carbon skeleton and ring-opening of the bridged ring via C–O bond cleav ... - RSC Advances (RSC Publishing) DOI:10.1039/D2RA01315K](https://pubs.rsc.org/image/article/2022/RA/d2ra01315k/d2ra01315k-s4_hi-res.gif)
Highly efficient construction of an oxa-[3.2.1]octane-embedded 5–7–6 tricyclic carbon skeleton and ring-opening of the bridged ring via C–O bond cleav ... - RSC Advances (RSC Publishing) DOI:10.1039/D2RA01315K