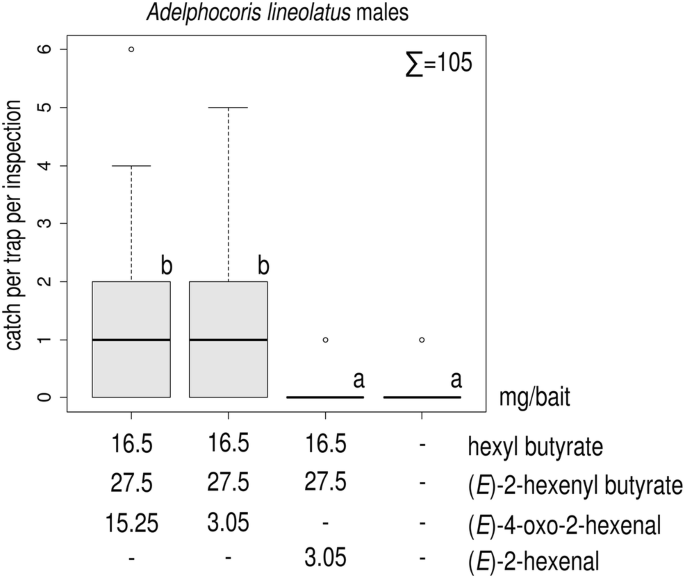
Sex Pheromone of the Alfalfa Plant Bug, Adelphocoris lineolatus: Pheromone Composition and Antagonistic Effect of 1-Hexanol (Hemiptera: Miridae) | SpringerLink

PDF) Synthetic- And DFT modelling studies on regioselective modified Mannich reactions of hydroxy-KYNA derivatives
Evaluation of the Antioxidant Activity of Cis/Trans-N-Phenyl-1,4,4a,5,8,8a-Hexahydro-3, 1-Benzoxazin-2-Imines

IJMS | Free Full-Text | Synthesis of Indole-Coupled KYNA Derivatives via C–N Bond Cleavage of Mannich Bases
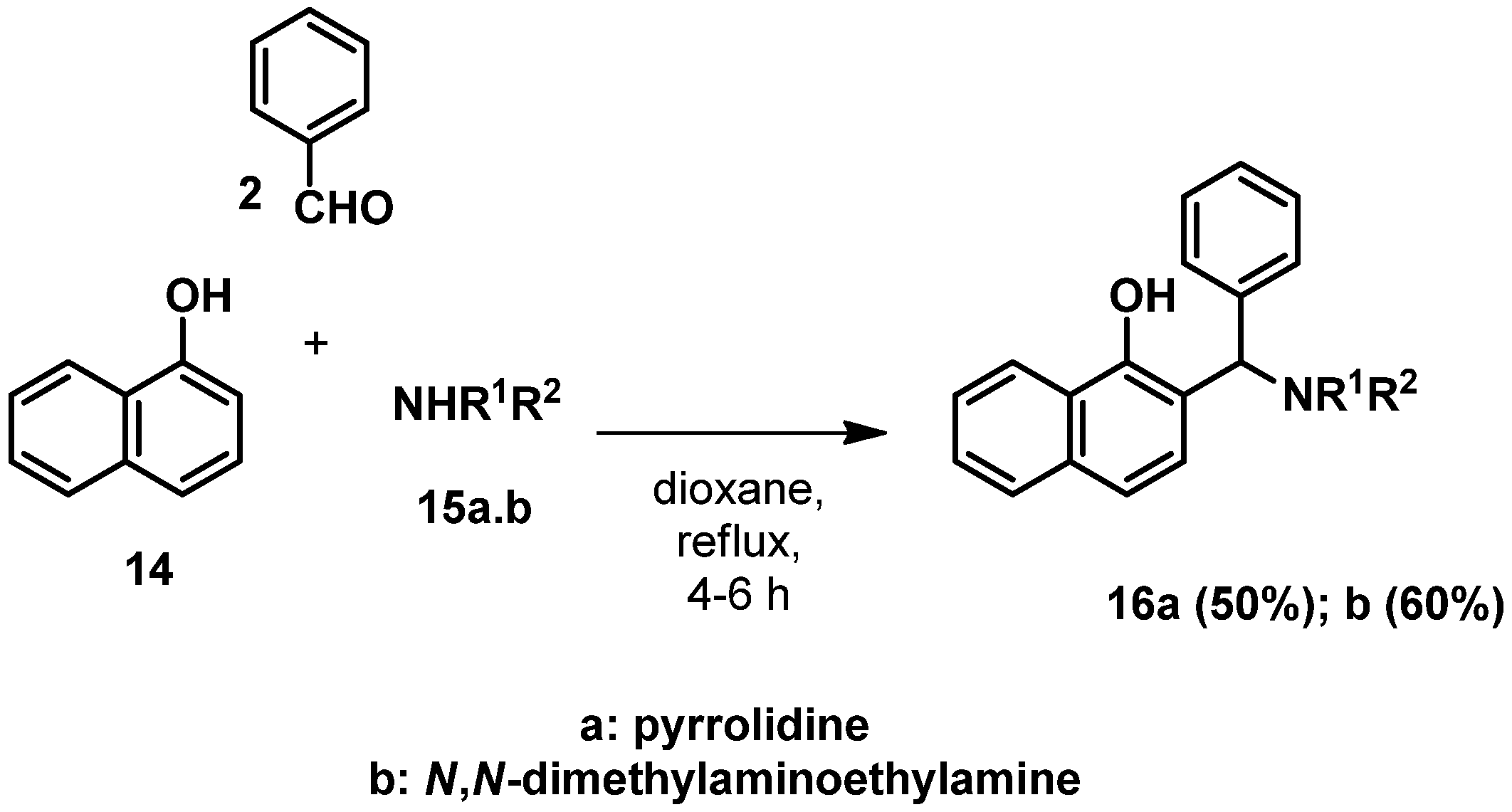
IJMS | Free Full-Text | Synthesis of Indole-Coupled KYNA Derivatives via C–N Bond Cleavage of Mannich Bases

Plants | Free Full-Text | Jacaranone Derivatives with Antiproliferative Activity from Crepis pulchra and Relevance of This Group of Plant Metabolites

Selective Synthesis of Fluorine‐Containing Cyclic β‐Amino Acid Scaffolds - Kiss - 2018 - The Chemical Record - Wiley Online Library
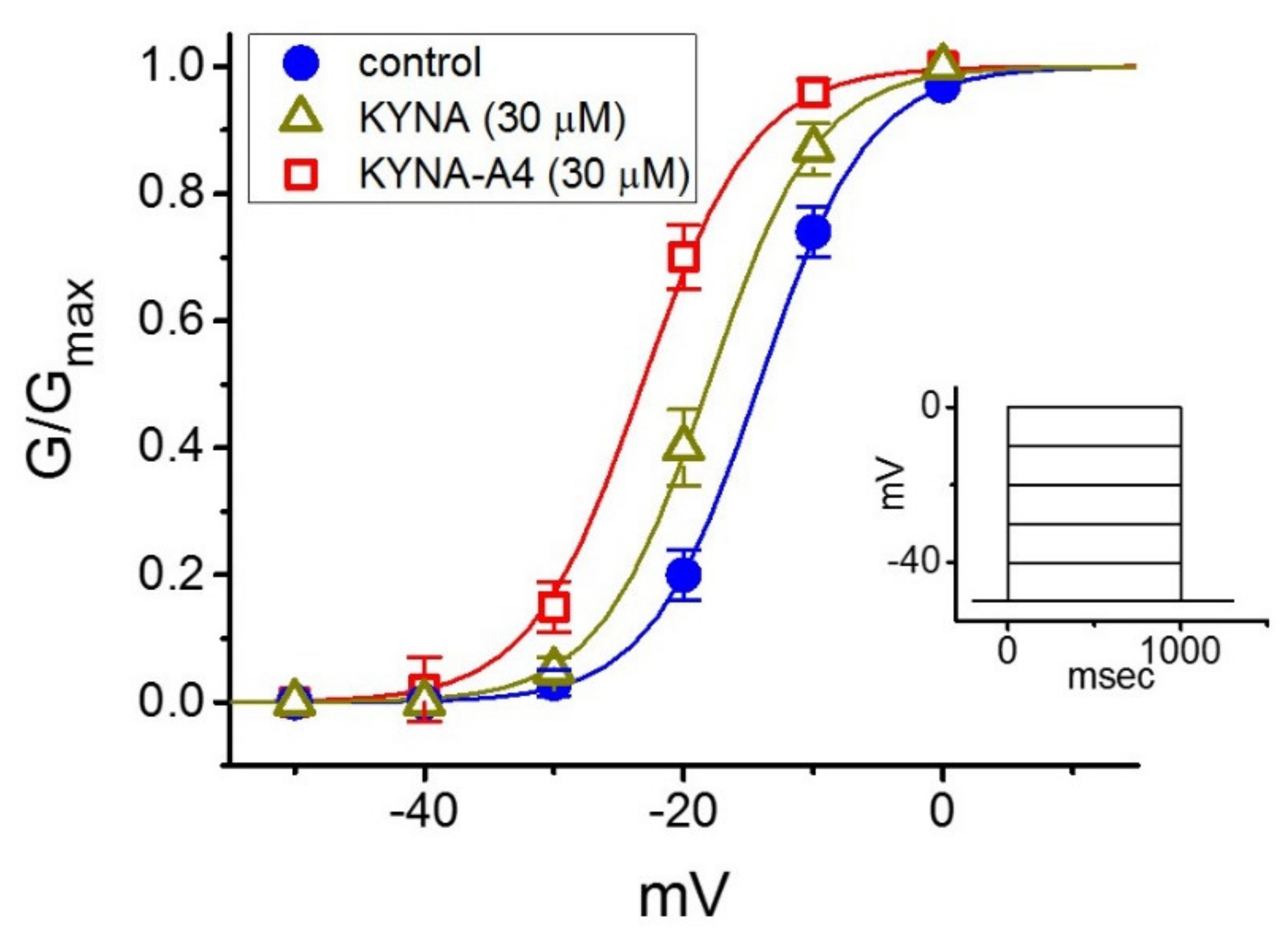
IJMS | Free Full-Text | Effective Activation by Kynurenic Acid and Its Aminoalkylated Derivatives on M-Type K+ Current

Fluorine‐Containing Functionalized Cyclopentene Scaffolds Through Ring Contraction and Deoxofluorination of Various Substituted Cyclohexenes - Remete - 2018 - European Journal of Organic Chemistry - Wiley Online Library

Selective Synthesis of Fluorine‐Containing Cyclic β‐Amino Acid Scaffolds - Kiss - 2018 - The Chemical Record - Wiley Online Library

Selective Synthesis of Fluorine‐Containing Cyclic β‐Amino Acid Scaffolds - Kiss - 2018 - The Chemical Record - Wiley Online Library
![Retro Diels Alder protocol for regioselective synthesis of novel [1,2,4 ]triazolo[4,3- a ]pyrimidin-7(1 H )-ones - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04345A Retro Diels Alder protocol for regioselective synthesis of novel [1,2,4 ]triazolo[4,3- a ]pyrimidin-7(1 H )-ones - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04345A](https://pubs.rsc.org/image/article/2020/RA/d0ra04345a/d0ra04345a-s1_hi-res.gif)
Retro Diels Alder protocol for regioselective synthesis of novel [1,2,4 ]triazolo[4,3- a ]pyrimidin-7(1 H )-ones - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04345A
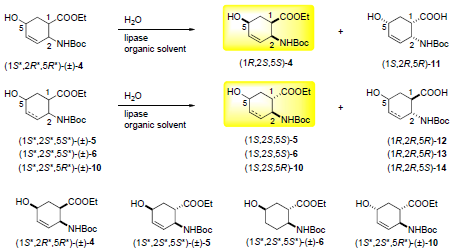
Molecules | Free Full-Text | Direct Enzymatic Route for the Preparation of Novel Enantiomerically Enriched Hydroxylated β-Amino Ester Stereoisomers

Recent Progress in Aryltrifluoromethylation Reactions of Carbon‐Carbon Multiple Bonds - Remete - 2022 - Chemistry – An Asian Journal - Wiley Online Library
![Retro Diels Alder protocol for regioselective synthesis of novel [1,2,4 ]triazolo[4,3- a ]pyrimidin-7(1 H )-ones - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04345A Retro Diels Alder protocol for regioselective synthesis of novel [1,2,4 ]triazolo[4,3- a ]pyrimidin-7(1 H )-ones - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04345A](https://pubs.rsc.org/image/article/2020/RA/d0ra04345a/d0ra04345a-s2_hi-res.gif)
Retro Diels Alder protocol for regioselective synthesis of novel [1,2,4 ]triazolo[4,3- a ]pyrimidin-7(1 H )-ones - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04345A









